Critique Review on Serotonin Engages an Anxiety and Fear-promoting Circuit in the Extended Amygdala
Abstract
Previous findings have proposed that drugs targeting 5-HT2C receptors could exist promising candidates in the treatment of trauma- and stress-related disorders. However, the reduction of conditioned freezing observed in 5-HT2C receptor knock-out (KO) mice in previous studies could alternatively be accounted for by increased locomotor action. To neutralize the derange of individual differences in locomotor activity, we measured a ratio of fright responses during versus before the presentation of a conditioned stimulus previously paired with a footshock (as a fear measure) by utilizing a conditioned licking suppression epitome. We first confirmed that v-HT2C receptor gene KO attenuated fear responses to distinct types of single conditioned stimuli (context or tone) independently of locomotor activity. We so assessed the effects of 5-HT2C receptor gene KO on chemical compound fear responses by examining mice that were jointly conditioned to a context and a tone and later re-exposed separately to each. We institute that dissever re-exposure to individual components of a complex fear memory (i.e., context and tone) failed to elicit contextual fear extinction in both 5-HT2C receptor factor KO and wild-blazon mice, and also abolished differences between genotypes in tone-cued fright extinction. This written report delineates a previously overlooked role of 5-HT2C receptors in conditioned fear responses, and invites circumspection in the time to come assessment of molecular targets and candidate therapies for the handling of PTSD.
Introduction
Posttraumatic stress disorder (PTSD), a trauma- and stress-related disorder, develops afterwards experiencing a traumatic event. With a worldwide lifetime prevalence of three.9% [1], PTSD has become a global health issue and is characterized by excessive physiological arousal and dysregulated fearfulness responses to contexts and cues previously associated with the traumatic event. Selective serotonin reuptake inhibitors (SSRIs) alleviate these symptoms partially, although approximately 40% of SSRI-treated patients are non-responders. Serotonin five-HT2C receptor gene KO in mice or 5-HT2C receptor adversary administration have been constitute to reduce feet-related behaviors [ii,3,iv,5,6], suggesting that serotonin five-HT2C receptors could be a promising target in the treatment of PTSD.
Pavlovian fright workout has been extensively used as a relevant model of PTSD [7]. Intraperitoneal (i.p.) injection of the v-HT2C receptor antagonist SB242085 (0.3 mg/kg) in rats reduced freezing behavior in contextual fear conditioning [6]. Consistent with Ohyama et al. (2016) [six], we recently assessed the freezing behavior of five-HT2C receptor KO mice in contextual fear conditioning and found that they exhibited a rapid within-session extinction, although there was no difference in retrieval and between-session extinction of fear memory compared to WT control mice [4]. Notwithstanding, freezing behavior, which is divers as the absence of movement except for respiratory-related movements [seven], can be affected past altered levels of locomotor activity. We take recently shown that 5-HT2C receptor KO mice display an increased locomotor action in both zero-maze and open-field tasks [4]. The reduction of freezing in 5-HT2C receptor KO mice or mice treated with 5-HT2C receptor antagonists could thus alternatively be explained by their higher locomotor action trait.
Moreover, according to associative learning theories, the intensity of fright responses to stimuli tin be affected when multiple conditioned stimuli (CSs) previously paired with the same aversive upshot interact with each other [8,9,10,11]. Indeed, it has been shown that a reduction in fright responses to a cue is associated with sustained contextual fear responses when cue-specific and contextual information are combined during conditioning but separately presented during testing [12,13,14]. Underestimating cue-competition effects may thus pb to an incomplete evaluation of potential PTSD therapeutics, such as those targeting 5-HT2C receptors.
The aim of the electric current set of experiments was twofold. We first attempted to neutralize the confounding gene of locomotor activity while assessing the effect of 5-HT2C receptor gene KO in fear responses. Second, we aimed to assess the role of 5-HT2C receptors in fear responses to a unmarried CS (i.eastward., the context in Experiment 1; a tone in Experiment ii) paired with a foot shock as an aversive unconditioned stimulus (U.s.a.), or to 2 CSs (context + tone in Experiment 3) paired together with the The states but extinguished in separate sessions. In Experiment ane and ii, we used a conditioned lick suppression procedure where water-deprived mice are presented with a single CS (the context or a tone) previously paired with a US, while they consumed water. Retrieval of the CS-Us fear memory is expected to suppress water consumption, thereby reducing the number of licks during the CS presentation. We calculated a ratio of the number of licks before versus during the CS presentation, thereby neutralizing the potential issue of locomotor activity in fear responses. In Experiment 3, cue-specific (i.e., tone) and contextual information were embedded during conditioning merely extinguished separately to test whether this process affects fear responses. In other words, we tested whether five-HT2C receptor cistron KO is still effective in the compound state of affairs.
Methods and materials
Animals
We used 5-HT2CR KO male mice (RRID:IMSR_JAX:015821) [fifteen, 16], C57BL/6 Northward WT male littermates, and C57BL/6 N WT male mice supplied from Nihon SLC Co. Ltd (Hamamatsu, Japan). 5-HT2C receptor KO mice and their littermates were backcrossed to the C57BL/half dozen Due north strain for more than than 10 generations. All mice were aged >56 days prior to the beginning of the experiment. Animals were kept in rooms under an alternating lite-dark cycle (calorie-free from 7 p.m. to vii a.chiliad.) at 25 ± 2 °C and a relative humidity of 40–60%. All experimental sessions were performed during the dark menses. The handling of animals complied with the Guidelines for the Care and Use of Laboratory Animals of the Fauna Research Commission of Hokkaido University, and all procedures were canonical by the Beast Research Committee of Hokkaido University (approval no. xviii-0070).
Behavioral procedures
Appliance
All experiments were conducted in 2 identical modular chambers (lab-hacks.com, France, 18 × 28 × 31 cm3) containing a 3 cm-wide mag illuminated past a warm white LED at 350 mcd of typical luminous intensity (Adafruit LED Sequins). Chambers were composed of a grid floor made from stainless steel rods separated by a 0.half-dozen cm gap. The floor and ceiling features could exist manipulated to create a second distinct context (Context B). In Context B, the roof was of triangular shape and the filigree floor was fully covered past a white plastic console with a rough surface (Fig. 1). In Context A, the triangular roof and white plastic floor were removed. Foot shocks could be delivered to the filigree floor past a precision animal shocker (H13-15, Coulbourne Instruments, Harvard Apparatus). The human foot shocks, ii speakers, and the LED were controlled by a microcontroller (Arduino Mega2560, Arduino, Italy). A 12 kHz pure tone could exist delivered via the 2 speakers at 80 dB (C-scale). Magazine entries were detected by an infrared intermission beam sensor (Adafruit, IR Break Beam Sensor – five mm LEDs) placed at the entrance of the mag around the water port level acme and were continued to the microcontroller. The number of licks were detected during the acclimation and contextual fear extinction phases by the microcontroller using a sensor (MPR121-12, Adafruit, Usa, NY) connected to the spout of the water bottle via a candy wire. All data were sent from the microcontroller to a microcomputer (Raspberry Pi, Uk) through a USB cable. A custom-written python script was used to collect and save data in a comma-separated values (CSV) file.
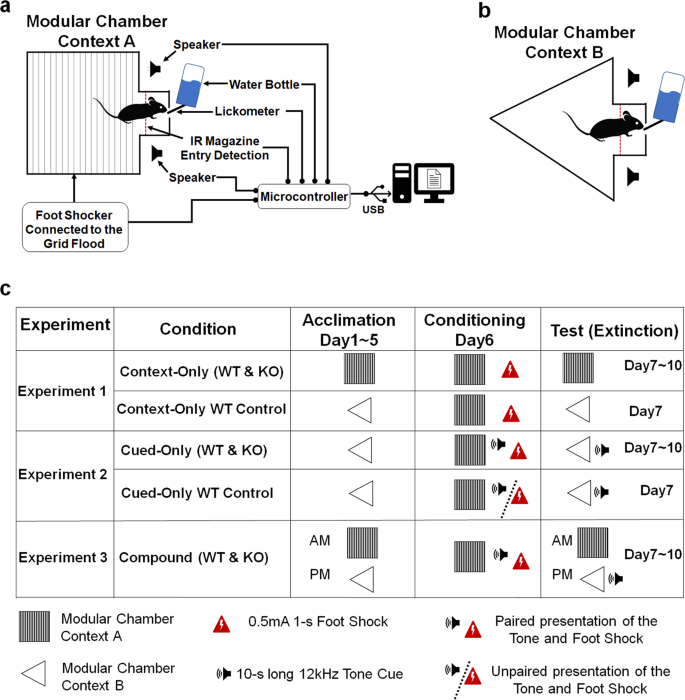
a Schematic cartoon of a mouse drinking water inside the magazine of a modular chamber (Context B). The foot daze output was connected to the grid floor. The foot shock input, 2 speakers, the infrared (IR) mag entry sensor, and the lickometer were continued to a microcontroller. The microcontroller was connected to a microcomputer via USB cable. b Schematic representation of the modular chamber arranged in Context A, with a white plastic cream canvas roofing the grid floor and a triangular-shaped transparent acrylic ceiling. c Experimental flowchart for each condition (in Rows). Columns represents the iii different phases of the procedure (Acclimation, Conditioning, and Test).
Experiment 1: Contextual conditioned lick suppression
After mice were handled for five min for at to the lowest degree 3 sequent days, water bottles were removed from their home cage 24 h prior to the starting time of the experiment. The next day, mice in the Context-Only condition received 5 days (Day 1-5) of acclimation to the workout chambers (in Context A for WT and KO mice; in Context B for WT Control mice) (Fig. 1). Five-24-hour interval acclimation was used to ensure that mice reached a steady rate of licking operation (Supplementary Fig. 1). During this stage, mice had free access to the water-filled lick tubes. Mice were given free access to water for 30 min in their home cage after each session throughout the whole experiment. Nosotros excluded whatever mouse that failed to drink from the water-filled lick tubes on Days 1 and 2. On Day 6, all mice in the Context-Only condition were presented half dozen times with a one-s, 0.five-mA foot shock in Context A. Magazine entries and access to the water-filled lick tubes were prevented by an opaque black plastic sheet placed in front end of the magazine. The onsets of the shock occurred at 5.xv, 9.xv, 13.15, 17.15, 21.15, and 25.15 min into the session. On Days 7 to 10, WT and KO mice in the Context-But condition were tested for contextual conditioned lick suppression in Context A, while WT Control mice in the same status were tested only on Day seven in Context B. To validate our procedure, WT Control mice were acclimated and tested in Context B, while they received contextual fearfulness conditioning in Context A on Day half-dozen. Nosotros expected that fear responses of WT mice would be higher than that of WT Control mice, which would suggest that fear responses of WT mice are a consequence of the retrieval of the Context A-foot shock memory. The absence (or at least low level) of fright responses in WT Control mice would also ensure that WT mice performance is not due to contextual fearfulness generalization (i.eastward., WT mice fear responses are Context A-specific). A contextual conditioned lick suppression test session lasted 30 min and was similar in every aspect to an acclimation session.
Experiment 2: Cued conditioned lick suppression
The acclimation phase for mice in the Cued-Only status was similar in every attribute to that of WT Control mice in the Context-Only condition. On Twenty-four hours vi, WT and KO mice in the Cue-Only condition were presented six times with a 10-south long 12 kHz pure tone cue co-terminated with a 1-s 0.5-mA pes shock in Context A. For WT Control mice in the aforementioned condition, the half dozen presentations of the tone cue and the foot shock were unpaired. Magazine entries and access to the water-filled lick tubes were prevented past an opaque black plastic sheet placed in front of the magazine. The onsets of the tone occurred at 5, ix, 13, 17, 21, and 25 min into the session for WT and KO mice. For WT Control mice, the onsets of the foot stupor occurred at 8.v, xi.7, 15.9, xviii.3, 22.25, and 23.8 min into the session. On Days 7 to 10, WT and KO mice in the Cue-But status were tested for cued conditioned lick suppression, while WT Control mice in the same status were tested only on Twenty-four hours seven. To validate our procedure, WT Command mice received unpaired presentations of the tone and the pes shock during the cued fearfulness workout phase on Day six, while the number of presentations of the tone and foot shock was the aforementioned as for WT mice. We expected that fear responses of WT mice would exist higher than that of WT Command mice, which would suggest that fear responses of WT mice are a effect of the retrieval of the tone-foot shock retentivity. The absence (or at to the lowest degree low level) of fear responses in WT Control mice would also ensure that WT mice learned the predictive value of the tone, due to the temporal contiguity of the tone and the pes shock during the conditioning phase. For all mice in the Cued-But condition, a cued conditioned lick suppression test consisted of a thirty min session in Context B, during which five cumulative seconds within the magazine triggered the 10-s tone cue alone. Later the offset of the latter, five cumulative seconds inside the magazine triggered the tone cue again, up to 5 times. The examination session ended if 5 tone cues were triggered, or after xxx min elapsed.
Experiment 3: Chemical compound context-cue conditioned lick suppression
To examine the effects of cue competition in our paradigm, we used a chemical compound of two CSs (context and tone) paired together with a foot stupor, just extinguished in split sessions. All mice in the Compound status received two sessions of acclimation per day. In the morning, the acclimation session was the same every bit that of WT and KO mice in the Context-Only status. In the afternoon, the 2nd acclimation session was the same equally that of WT Command mice in the Context-Only condition. Twenty-four hour period half-dozen for WT and KO mice in the Chemical compound condition was like in every aspect to that of WT and KO mice in the Cued-Just condition. On Days vii to 10, all mice in the Chemical compound condition were tested for contextual conditioned lick suppression in the morning, and cued conditioned lick suppression in the afternoon (6 h later). The contextual conditioned lick suppression test consisted of the aforementioned test procedure as that of WT and KO mice in the Context-Only status, and the cued conditioned lick suppression test was similar in every attribute to the exam process used in WT and KO mice in the Cued-Only status.
Data analysis
To neutralize individual differences in locomotor action and water consumption, we calculated a conditioned lick suppression ratio [17] as a measure out of fear response calculated as follows:
$${{{\mathrm{Conditioned}}}}\;{{{\mathrm{Lick}}}}\;{{{\mathrm{Suppression}}}}\;{{{\mathrm{Ratio}}}} = \frac{{{{{\mathrm{Baseline}}}} - {{{\mathrm{Performance}}}}}}{{{{{\mathrm{Baseline}}}} + {{{\mathrm{Operation}}}}}}$$
For the contextual conditioned lick suppression ratio, the Baseline score refers to averaged rates of licks measured during the 30-min sessions of the two last days of the acclimation phase (i.e., Days 4 and five), and the Performance score refers to the rates of licks measured during the thirty-min sessions of the test stage (i.e., Days seven to 10).
For the cued conditioned lick suppression ratio, the Baseline score refers to rates of licks during the 5-s menstruation that precedes the onset of the tone cue, and the Performance score refers to rates of licks during the first 5 south of the tone cue. A low or negative score indicates a low or an absence of conditioned lick suppression (i.e., conditioned fear response), respectively, while a high score indicates a strong conditioned lick suppression.
Data were analyzed using SPSS 23.0 (IBM, NY, USA) and plotted with Prism (GraphPad, San Diego, USA). The sample size (N) was decided based on previous reports using the same type of experiments [18, 19]. No randomization was used to make up one's mind how animals were allocated to experimental groups. No blinding was done because all the information were collected automatically. An unpaired Student's t-exam for contained samples was used for ii-group comparisons, with Bonferroni correction when required. A 2-style analysis of variance (ANOVA) for repeated measures followed by the Bonferroni postal service-hoc test was used for multiple comparisons. If Mauchly's sphericity exam was significant, a Greenhouse-Geisser correction was used. A log-rang (Mantel-Cox) test was used when comparing the survival rate performance betwixt groups in the cued conditioned lick suppression task. Data are expressed every bit means ± SEM, except for the survival rate where probabilities of survival are presented.
Results
Experiment 1: five-HT2C receptor KO mice displayed reduced contextual conditioned fear responses compared to WT mice
When tested in the same context where contextual fear workout was conducted (Context B), KO mice (Context-Only KO Group, N = 11) showed a significantly lower conditioned lick suppression ratio when compared to WT mice (Context-Only WT Group, Due north = eleven) (Fig. 2a, b) as revealed by a 2-manner ANOVA for repeated measures (F(1, x) = vii.369, p = 0.022). Mice of both genotypes extinguished contextual fear responses beyond sessions of the test phase, as suggested past a statistically significant decrease in conditioned lick suppression ratio across extinction sessions (F(one.539, 15.390) = 5.219, p = 0.005, with a Greenhouse-Geisser correction). No statistically significant interaction between groups and extinction days was found (F(one.603, 16.030) = 0.720, p = 0.473, with a Greenhouse-Geisser correction). These information indicate that KO mice manifested reduced contextual fear responses compared to WT mice, but extinguished their fearfulness responses at the aforementioned rate.
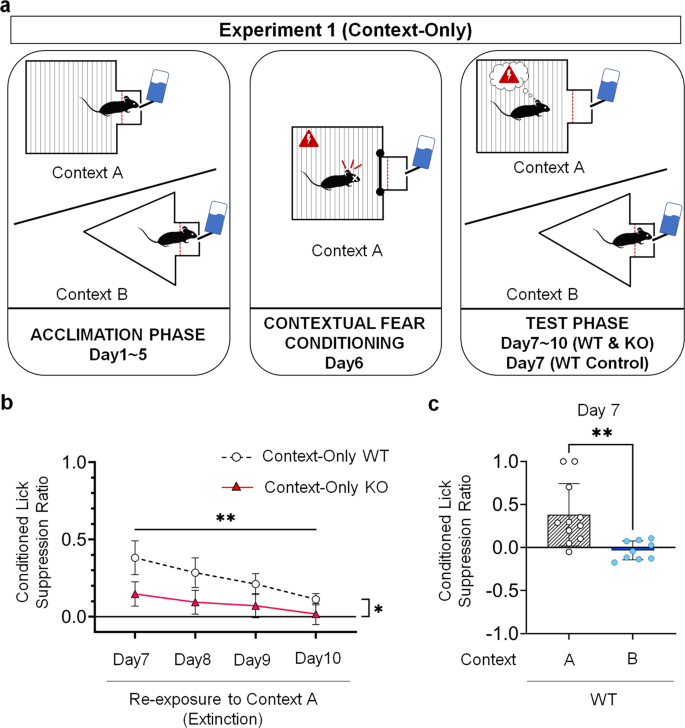
a Schematic cartoon of each of the three phases of the contextual condition lick suppression task. b Contextual conditioned lick suppression performance in WT mice (Context-Only WT Grouping; Northward = 11) compared to KO mice (Context-Only KO Group; Northward = 11) tested from Day 7 to Day 10 in the same context (Context A) where they received the fear workout session. Two-way analysis of variance (ANOVA) for repeated measures, followed past Bonferroni post-hoc test. Information are expressed as means and error bars represent ±SEM. Asterisks indicate P-values (*P < 0.05, **P < 0.01). c Conditioned lick suppression performance in WT mice tested at 24-hour interval 7 in the same context every bit the conditioning context (Context-Only WT Group; Northward = 11) compared to WT mice tested at Twenty-four hour period 7 in a different context (Context B) than the conditioning context (Context-Merely Control WT Group; N = 9). Two-tailed t-exam for independent samples with a Bonferroni correction. Data are expressed every bit means and mistake bars correspond ±SEM. Asterisks signal P-values (**P < 0.01).
Every bit a validation of the contextual conditioned lick suppression job used here, we ran an boosted group of WT mice (Context-Merely Control WT, N = 9) acclimated and tested in a context (Context A) that differs from that in which they received contextual fear conditioning (Context B). Their functioning on the first 24-hour interval of extinction (Twenty-four hour period seven) was compared with that of mice from the Context-Merely WT Group on the aforementioned day. The higher ratio observed in the Context-Only WT Grouping (Mean = 0.3810, SD = 0.3621) compared to the Context-Merely Control WT Grouping (Mean = −0.0334, SD = 0.1093) was statistically significant as determined past a two-tailed t-test for independent samples with a Bonferroni correction (t(12.162) = three.601, p = 0.004) (Fig. 2c). These results confirmed that the conditioned fearfulness responses observed in the Context-Merely WT Grouping were a result of the Context B→Foot shock memory formed during the contextual fear workout phase and retrieved during the test stage. These data validate the employ of contextual conditioned lick suppression every bit a measure of fright responses to a context previously paired with an aversive consequence.
Experiment 2: 5-HT2C receptor KO mice display reduced cued conditioned fear responses compared to WT mice
We first compared the cued conditioned lick suppression ratio of mice during the first tone delivery at Day 7 (i.e., Extinction Day 1) and constitute a lower suppression ratio in the Cue-Only KO Group (N = 8, Mean = 0.7968, SD = 0.0674) compared to the Cue-Simply WT group (N = 9, Mean = 0.9706, SD = 0.0470), an outcome that was statistically significant as determined past a two-tailed t-exam for independent samples with a Bonferroni correction (t(15) = half-dozen.228, p < 0.001) (Fig. 3b). These data indicate that KO mice manifest reduced cued fear responses compared to WT mice during the starting time tone presentation.
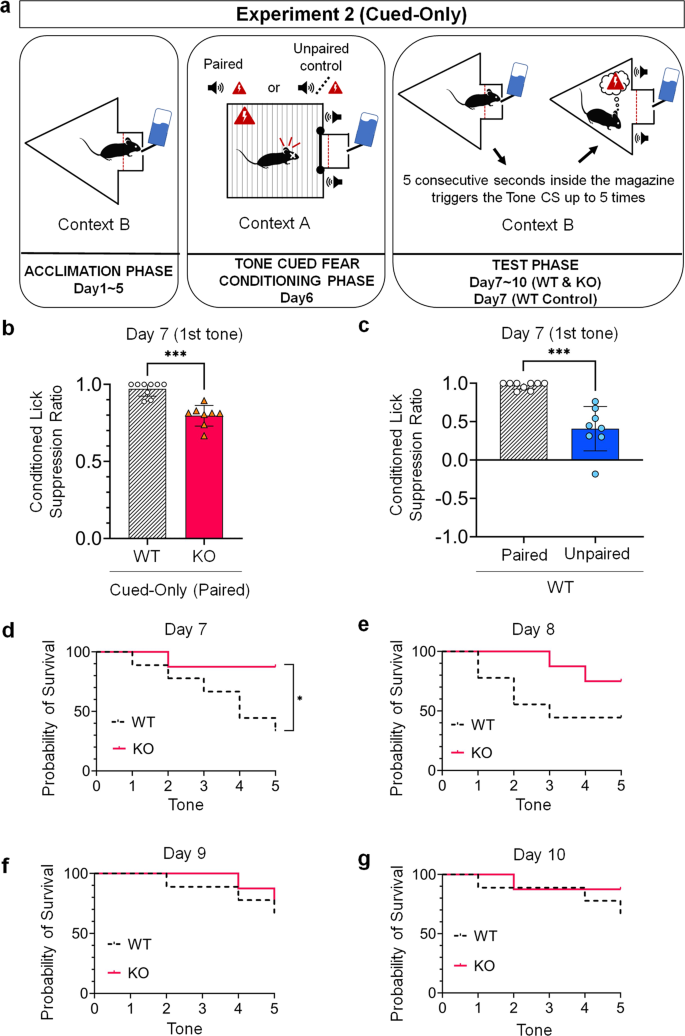
a Schematic drawing of each of the three phases of the cued status lick suppression task. b Cued conditioned lick suppression performance in WT mice (Cued-Only WT Group; Northward = 9) compared to KO mice (Cued-Only KO Grouping; Northward = 8) during the beginning tone delivery at Day seven, in a different context (Context B) from that of the conditioning phase (Context A). c Cued conditioned lick suppression performance during the first tone commitment at 24-hour interval 7 in WT mice that received paired presentations of the tone and footshock during the conditioning phase (Cued-Only WT Group; Northward = 9) compared to WT mice that received unpaired presentation of the tone and footshock during the conditioning phase (Cued-But WT Control Group; North = 8). Both groups where conditioned in Context A and tested in Context B. Two-tailed t-test for independent samples with a Bonferroni correction. Data are represented every bit means and error bars represent ±SEM. Asterisks indicate P-values (***P < 0.001). d–g Percentage probability of survival charge per unit between WT (Cued-Simply WT Grouping; N = 9) and KO mice (Cued-Only KO Group; N = 8) tested from Solar day 7 to Twenty-four hour period 10. Log-rank (Mantel-Cox) tests. Asterisks indicate P-values (*P < 0.05).
As a validation of the contextual cued lick suppression task used here, nosotros ran an additional group of WT mice (Cue-But Command WT, N = eight) similar in every aspect to mice from the Cue-Only WT Group, except for the conditioning phase where mice from the Cue-Simply Control WT Grouping received unpaired presentations of the tone and pes shock. We plant a significantly lower cue-conditioned suppression lick ratio in mice from the Cue-Only Control WT mice (Mean = 0.4087, SD = 0.2887) compared to mice from the Cue-Simply WT Grouping (Mean = 0.9706, SD = 0.0470) as determined past a 2-tailed t-test for contained samples with a Bonferroni correction (t(7.330) = v.440, p = 0.001) (Fig. 3c). This effect validates the use of cued lick suppression as a measure of fear responses to a tone cue previously paired with an aversive event. It is worth noting that all mice from the Cue-Just Command WT Group triggered the 5 tones during the test session (i.eastward., 100% of probability of survival; data not shown).
To further clarify cued fright responses inside each extinction session, we compared the per centum probability of survival rate between genotypes and constitute a significantly lower score in mice from the Cue-Merely WT Grouping compared to mice from Cue-Merely KO Group but on the starting time day of extinction (Day vii: χ 2 = iv.36, p = 0.037; Twenty-four hour period 8: χ 2 = ii.21, p = 0.014; 24-hour interval 9: χ 2 = 0.19, p = 0.66; Day x: χ 2 = 0.88, p = 0.35) (Fig. 3d–g). These results further strengthen the conclusion that KO mice manifest reduced fear responses to the tone cue, as they were less impacted past information technology compared to WT mice.
During cued conditioned lick suppression tests, five consecutive seconds spent inside the mag triggered the delivery of the tone cue. The latter could exist triggered upward to 5 times each session. Thus, mice were naïve to these contingencies only for the first tone delivery at Solar day 7 (i.e., Extinction Mean solar day i), which consists of a Pavlovian conditioning examination. However, mice might have learned along with subsequent trials that their behavior (i.east., entering the magazine for 5 cumulative seconds) triggers the delivery of a tone previously paired with a shock (Fig. 3a). As a outcome, we tin can expect that mice learn to avert entering the magazine at the early on stages of the test stage and outset entering the magazine again as the extinction procedure progresses, consistent with the percentage probability survival charge per unit (Fig. 3d–k). In addition to the survival charge per unit, we examined cued fear extinction for each solar day by averaging the conditioned lick suppression ratios measured for all tones triggered. As mice did non always trigger the five tones, nosotros used a mixed-effects assay of variance (ANOVA) and found that mice of both genotypes extinguished cued fearfulness responses across sessions of the test stage, as suggested past a statistically significant decrease in conditioned lick suppression ratio across extinction sessions (F(3, 24) = seven.22, p = 0.001) (Supplementary Fig. 2). No statistically pregnant difference between genotype or no statistically significant interaction between groups and extinction days was plant.
Experiment 3a: separate re-exposure to cue- and context-conditioned stimuli prevents contextual fright extinction in five-HT2C receptor KO and WT mice and abolishes genotype differences in contextual fright responses
We further examined whether 5-HT2C receptor gene KO exerts the same effects on fear responses even when re-exposing cued and contextual information separately (Fig. 4a). The assay of re-exposure of contextual information using a two-manner ANOVA for repeated measures revealed no statistically significant deviation between WT (Compound-Context WT Group, N = 7) and KO mice (Compound-Context KO Group, Northward = 7) in the Chemical compound status (F(1.000, 6.000) = 0.288, p = 0.611), and that the conditioned suppression ratio did not differ significantly betwixt extinction days (F(2.014, 12.083) = 1.508, p = 0.260) (Fig. 4b). At that place was no statistically meaning genotype*extinction interaction (Greenhouse-Geisser correction) (F(3, 18) = 0. 492, p = 0.692). These data indicate that both KO and WT manifested a similar level of contextual conditioned lick suppression and highlight a deficit of contextual fear extinction in the Compound condition.
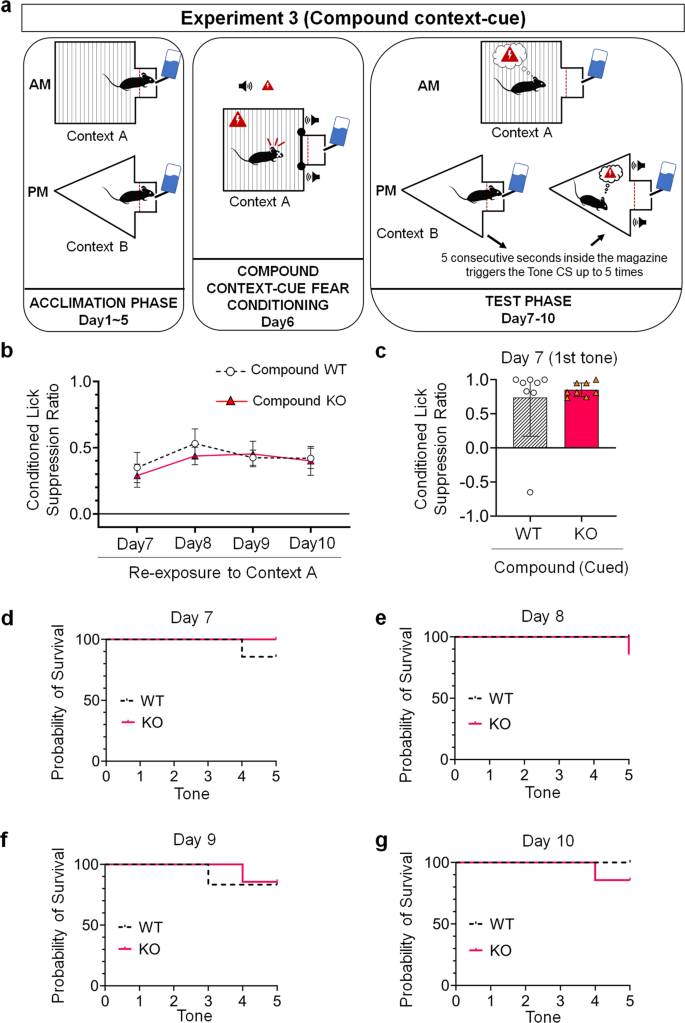
a Schematic drawing of each of the iii phases of the compound context-cue status lick suppression task. b Contextual conditioned lick suppression performance in WT mice (Compound WT Group; N = 7) compared to KO mice (Compound KO Grouping; N = 7) tested from Day seven to Day x in the same context (Context A) where they received the fearfulness conditioning session. Two-fashion analysis of variance (ANOVA) for repeated measures. Data are represented as ways and fault confined represent ±SEM. c Cued conditioned lick suppression performance in WT mice (Compound WT Group; N = seven) compared to KO mice (Compound KO Group; Due north = 7) during the first tone delivery at 24-hour interval 7 in a different context (Context B) from that of the workout stage (Context A). Ii-tailed t-test for contained samples. Black full lines and dashed black lines of violin plots stand for medians and quartiles (first and tertiary) respectively. d–chiliad Percentage probability of survival rate between WT (Chemical compound WT Group; N = 7) and KO mice (Compound KO Group; N = 7) tested from Day 7 to Day 10. Log-rank (Mantel-Cox) tests.
Experiment 3b: compound context-cue fear conditioning abolishes differences in cued fear responses in 5-HT2C receptor KO and WT mice
Re-exposing mice to cued data also resulted in a different consequence. We first compared the cued conditioned lick suppression ratio of mice during the offset tone delivery at Day seven (i.e., Extinction Day one) and constitute that the conditioned lick suppression ratio in the Chemical compound-Cue KO Group (N = seven, Hateful = 0.8376, SD = 0.1003) compared to that of the Compound-Cue WT group (North = vii, Hateful = 0.7240, SD = 0.6098) was not different as determined by a two-tailed t-test for independent samples (t(12) = −0.486, p = 0.636) (Fig. 4c). These information indicate that both genotypes manifested similar cued fear responses during the showtime tone presentation in the Compound status.
To further analyze cued fearfulness responses within each extinction session, we compared the percentage probability of survival rate betwixt genotypes and did not find whatever differences between the Compound-Cue WT and Compound-Cue KO Groups for each 24-hour interval of the examination phase (Mean solar day seven: χ 2 = 1.00, p = 0.32; Twenty-four hours eight: χ two = 2.17, p = 0.xiv; Day nine: χ ii = 0.003, p = 0.96; Day x: χ 2 = 1.00, p = 0.32) (Fig. 4d–g). These results further strengthen the decision that both genotypes manifested similar fear responses to the tone cue.
In addition to the survival charge per unit, we examined cued fear extinction for each day past averaging the conditioned lick suppression ratios measured for all tones triggered. As mice did not e'er trigger the five tones, we used a mixed-furnishings assay of variance (ANOVA) and constitute that mice of both genotypes extinguished cued fear responses across sessions of the test phase, as suggested by a statistically significant subtract in conditioned lick suppression ratio across extinction sessions (F(3, 35) = 4. 71, p = 0.007) (Supplementary Fig. 2). No statistically pregnant departure between genotypes and no statistically significant interaction between groups and extinction days were found.
Word
Prior reports showed a reduction of freezing responses in v-HT2C receptor factor KO mice [20] or afterwards 5-HT2C receptor antagonist assistants [6], but these findings could alternatively be explained by increased locomotor activity in these animals. To resolve this problem, we used a conditioned licking suppression image and clearly demonstrated that genetic deletion of five-HT2C receptors in mice attenuates fear responses in contextual or cued conditioning independent of changes in locomotor action. (Figs. 2 and 3). To our knowledge, the present study is the showtime to control for the effects of locomotor activity while assessing the function of 5-HT2C receptors in contextual fright responses. The procedures developed hither should bear witness useful in exploring potential therapeutics for fear-related symptoms, especially when using animal models with unlike baselines of locomotor activeness compared to WT animals.
Previous studies have demonstrated that the amygdala is necessary for cued fear while the hippocampus is necessary for contextual fearfulness [21] and that five-HT2C receptors are expressed in both regions [22, 23]. A previous study showed that stress increased the expression of 5-HT2C receptors in the amygdala, and the pharmacological occludent of 5-HT2C receptors in the amygdala attenuated fear responses to a tone cue previously paired with a footshock [24]. Thus, it is likely that the reduction of cued fright response in 5-HT2C receptor KO mice (Fig. 3) is due to the lack of 5-HT2C receptors in the amygdala. In accordance with this hypothesis, our previous written report showed that the pharmacological blockade of five-HT2C receptors in the ventral hippocampus did not reduce contextual fear response in rats [25]. It should be noted that we injected the antagonist immediately before the re-exposure to examine the office of 5-HT2C receptors in the retrieval of contextual fearfulness memory. Moreover, nosotros previously found that 5-HT2C receptor KO mice displayed a slower acquisition of fearfulness conditioning [4]. Thus, it is possible that the reduction of contextual fearfulness response in 5-HT2C receptor KO mice (Fig. ii) is due to a deficit in the conquering of contextual fear conditioning rather than the retrieval of contextual fearfulness retention. Alternatively, 5-HT2C receptors in the dorsal hippocampus rather than the ventral hippocampus might play an essential part in the retrieval of contextual fright memory.
It should also exist noted that neither our licking suppression prototype nor conventional freezing paradigm can discriminate amidst the deficits in acquisition, consolidation, and retrieval of fear memory in specific cases. Although the conditioned licking suppression epitome excludes the confounding effects of locomotor activity, this paradigm cannot assess the process of fear acquisition because we cannot observe licking during fear conditioning. Thus, it is impossible to discriminate among the deficits in acquisition, consolidation, and retrieval of fear memory when significant differences in the number of licking between groups appear from the beginning of the test stage. The conventional freezing image can assess the procedure of fear acquisition, but it would exist difficult to interpret the results when pregnant differences in locomotor action betwixt groups are observed. Future studies should address these limitations. At to the lowest degree for a while, these two paradigms would be complementary to each other.
The reduction of contextual fear responses in five-HT2C receptor KO mice might exist due to increased BDNF levels in the hippocampus because a previous study demonstrated that BDNF expression in the hippocampus was increased in 5-HT2C receptor KO mice [xx]. BDNF Val66Met polymorphism has been associated with impaired memory part, vulnerability to stress, stress-related disorders [26], and cognitive/affective deficits in other psychiatric disorders such as schizophrenia [27]. Thus, it is possible that 5-HT2C receptor KO mice have the resilience to stress by increasing BDNF expression in the hippocampus. Supporting this, 5-HT2C receptor KO mice displayed anxiolytic phenotype [four]. However, the effects of BDNF on fear memory were brain region/timing-dependent and inconsistent among studies [26]. Further studies using conditional KO are required to analyze whether the increased BDNF expression is involved in the reduction of fear responses in v-HT2C receptor KO mice.
Surprisingly, in a procedure where contextual and cue-specific information where imbedded during conditioning, but tested separately, we found (i) that all differences observed in cued or contextual conditioning were abolished between 5-HT2C receptor KO and WT mice and (2) sustained contextual fearfulness responses over extinction trials. It is worth noting that five-HT2C receptor KO mice manifested a like level of contextual fearfulness responses compared to WT mice in this status, suggesting that the reduction of fright responses observed in five-HT2C receptor KO mice in the Context-Just condition is less likely to result from a lower sensitivity to the aversive The states, and/or a arrears in learning and memory processes. Our results are in line with previous studies in good for you human volunteers showing that a reduction in fear responding to a specific cue is associated with sustained contextual fearfulness responses when contextual and cue-specific data are combined during conditioning, simply tested separately [12,xiii,14]. Most importantly, the fact that this consequence was observed in both KO and WT mice suggests that the five-HT2C receptor is not necessary for contextual fear expression in our Chemical compound condition. This finding questions the relevance of targeting 5-HT2C receptors for developing therapeutics for PTSD, at to the lowest degree when it comes to exposure-based therapies, and urges caution in the cess of time to come candidate drugs.
How a potential interaction between contextual and cue-specific information led to sustained contextual fear responses remains speculative. Associative learning theories state that cue competition occurs when multiple cues are presented simultaneously and followed past a The states, a prediction that holds truthful between cue-specific and contextual data [25, 28]. According to this theoretical framework, cues with the highest saliency become the best predictors of the U.s.a., thus provoking the strongest CRs. This miracle is well documented in a wide variety of procedures and is referred to as overshadowing [29,30,31]. It is worth noting that the conditioning phase in both Cued-Only and Compound atmospheric condition is the aforementioned; that is, both tone-cue and contextual information are conditioned to the US. The chief difference betwixt these two conditions resides in the re-exposure of the tone-cue only (i.e., Cued-Only status) while tone-cue and contextual information are re-exposed in separate phases (i.eastward., Compound condition). This suggests that the condition of re-exposure post-obit the formation of a traumatic memory is a critical aspect in the effectiveness of re-exposure-based therapies to reduce fear responses.
We cannot exclude the possibility that the reduction of fearfulness responses in five-HT2C receptor KO mice observed in the Context-Only and Cued-Only conditions could effect from the absence of 5-HT2C receptor expression only during a critical period of development. Nonetheless, our results are consistent with a reduction of fear responses observed in adult rats administered intraperitoneally with a 5-HT2C receptor antagonist [6], and with the anxiolytic result of five-HT2C receptor antagonism in adult humans [32].
Based on the findings in the present study, we suggest 3 future directions. First, to amend the screening efficiency of candidate drugs, future studies testing potential therapeutics for PTSD should utilize a context-cue compound fear workout in addition to re-exposing contextual or cued fright-conditioned stimuli separately during extinction. 2d, nosotros suggest examining whether the cue-context interaction could be avoided when both elements are exposed simultaneously during the extinction stage. Virtual reality engineering appears to be an interesting strategy to tackle this event in humans and has a potential clinical translational value to patients [33]. Neutralizing context-cue interactions may assist in reconsidering the therapeutic efficacy of 5-HT2C receptor antagonists and other drugs that showed promising effects in preclinical studies simply failed to pass clinical trials. Third, our information invites mechanistic investigations at both behavioral and neural levels as to how contextual and cued fearfulness conditioning combined within the same procedure bypass anxiolytic-like effects such as those resulting from 5-HT2C receptor cistron KO in fear-related responses.
References
-
Koenen KC, Ratanatharathorn A, Ng L, McLaughlin KA, Bromet EJ, Stein DJ, et al. Posttraumatic stress disorder in the Globe Mental Wellness Surveys. Psychol Med. 2017;47:2260–74.
-
Martin CBP, Ramond F, Farrington DT, Aguiar Equally Jr, Chevarin C, Berthiau A-S, et al. RNA splicing and editing modulation of 5-HT2C receptor function: relevance to anxiety and aggression in VGV mice. Mol Psychiatry. 2013;xviii:656–65.
-
Marcinkiewcz CA, Mazzone CM, D'Agostino G, Halladay LR, Hardaway JA, DiBerto JF, et al. Serotonin engages an anxiety and fear-promoting excursion in the extended amygdala. Nature. 2016;537:97–101.
-
Nebuka Thousand, Ohmura Y, Izawa Southward, Bouchekioua Y, Nishitani N, Yoshida T, et al. Behavioral characteristics of v-HT2C receptor knockout mice: Locomotor activity, anxiety-, and fear memory-related behaviors. Behav Brain Res. 2020;379:112394.
-
Papp Yard, Litwa E, Gruca P, Mocaër E. Anxiolytic-like activity of agomelatine and melatonin in three beast models of anxiety. Behav Pharm. 2006;17:9–18.
-
Ohyama M, Kondo 1000, Yamauchi M, Imanishi T, Koyama T. Asenapine reduces feet-related behaviours in rat conditioned fear stress model. Acta Neuropsychiatr. 2016;28:327–36.
-
Fanselow, MS & Sterlace, SR in The Wiley Blackwell handbook of operant and classical workout 1st edn, (eds McSweeney, FK & Tater, ES) Ch. 6 (Wiley-Blackwell, New Jersey, 2014).
-
Rescorla, RA & Wagner, AR in Classical workout II: Electric current theory and research (eds Black, AH & Prokasy, WF) Ch. 3 (Appleton-Century-Crofts, New York, 1972).
-
Kamin LJ, Gaioni SJ. Compound conditioned emotional response conditioning with differentially salient elements in rats. J Comp Physiol Psychol. 1974;87:591–59.
-
Mackintosh NJ. Overshadowing and stimulus intensity. Anim Learn Behav. 1976;four:186–92.
-
Pearce JM, Hall One thousand. A model for Pavlovian workout: Variations in the effectiveness of conditioned but not of unconditioned stimuli. Psychol Rev. 1980;87:532–52.
-
Grillon C. Associative learning deficits increase symptoms of anxiety in humans. Biol Psychiatry. 2002;51:851–8.
-
Baas JMP, Van Ooijen L, Goudriaan A, Kenemans JL. Failure to condition to a cue is associated with sustained contextual fear. Acta Psychol (Amst). 2008;127:581–92.
-
Baas JM. Individual differences in predicting aversive events and modulating contextual anxiety in a context and cue conditioning paradigm. Biol Psychol. 2013;92:17–25.
-
Tecott LH, Sun LM, Akana SF, Strack AM, Lowenstein DH, Dallman MF, et al. Eating disorder and epilepsy in mice lacking 5-HT2c serotonin receptors. Nature. 1995;374:542–half-dozen.
-
Xu Y, Jones JE, Kohno D, Williams KW, Lee CE, Choi MJ, et al. five-HT2CRs expressed by pro-opiomelanocortin neurons regulate energy homeostasis. Neuron. 2008;sixty:582–nine.
-
Bouton ME, Bolles RC. Conditioned fearfulness assessed by freezing and by the suppression of 3 different baselines. Anim Learn Behav. 1980;viii:429–34.
-
Jozefowiez J, Witnauer JE, Miller RR. Two components of responding in Pavlovian lick suppression. Learn Behav. 2011;39:138–45.
-
Molet M, Miguez G, Cham HX, Miller RR. When does integration of independently caused temporal relationships take place? J Exp Psychol Anim Behav Process. 2012;38:369–80.
-
Hill RA, Murray SS, Halley PG, Binder MD, Martin SJ, van den Buuse Yard. Encephalon‐derived neurotrophic gene expression is increased in the hippocampus of 5‐HT2C receptor knockout mice. Hippocampus. 2011;21:434–45.
-
LeDoux JE. Emotion circuits in the brain. Annu Rev Neurosci. 2020;23:155–84.
-
Pompeiano K, Palacios JM, Mengod Thousand. Distribution of the serotonin five-HT2 receptor family mRNAs: comparison between five-HT2A and five-HT2C receptors. Mol Brain Res. 1994;163:136–40.
-
Li Q, Wichems CH, Ma L, Van de Kar LD, Garcia F, White potato DL Brain region-specific alterations of v-HT2A and 5-HT2C receptors in serotonin transporter knockout mice. J Neurochem (2003).
-
Ohmura Y, Yoshida T, Konno K, Minami 1000, Watanabe M, Yoshioka Grand Serotonin 5-HT7 Receptor in the Ventral Hippocampus Modulates the Retrieval of Fearfulness Retentivity and Stress-Induced Defecation. Int J Neuropsychopharmacol (2016).
-
Ayres JJB, Bombace JC, Shurtleff D, Vigorito M. Conditioned suppression tests of the context-blocking hypothesis: Testing in the absence of the preconditioned context. J Exp Psychol Anim Behav Process. 1985;11:1–14.
-
Notaras M, van den Buuse 1000. Neurobiology of BDNF in fearfulness memory, sensitivity to stress, and stress-related disorders. Mol Psychiatry. 2020;25:2251–74.
-
Notaras M, Colina R, van den Buuse MA. A role for the BDNF cistron Val66Met polymorphism in schizophrenia? A comprehensive review. Neurosci Biobehav Rev. 2015;51:15–xxx.
-
Urcelay GP, Miller RR. Two roles of the context in Pavlovian fright workout. J Exp Psychol Anim Behav Process. 2010;36:268–80.
-
Pavlov, IP (eds) Conditioned Reflexes: An Investigation of the Physiological Action of the Cerebral Cortex (Oxford Univ. Printing, London, 1927).
-
Kamin, LJ in Penalisation and aversive behavior 1st edn (eds Campbell, BA & Church, RM) Ch. 9 (Appleton-Century-Crofts, New York, 1969).
-
Mackintosh, NJ (eds) The Psychology of Animal Learning (Bookish Press, New York, 1974).
-
De Berardis D, Conti CM, Marini S, Ferri F, Iasevoli F, Valchera A, et al. Is in that location a part for agomelatine in the treatment of anxiety disorders? A review of published data. Int J Immunopathol Pharmacol. 2013;26:299–304.
-
Boeldt D, McMahon E, McFaul K, Greenleaf Due west. Using Virtual Reality Exposure Therapy to Enhance Treatment of Anxiety Disorders: Identifying Areas of Clinical Adoption and Potential Obstacles. Front end Psychiatry. 2019;x:773.
Acknowledgements
The authors thank JAM Post (https://www.jamp.com/index.cfm) for the English language review. We thank Shun Shimizu for his assist with information collection for the Context-Just condition. This piece of work was supported by JSPS KAKENHI grants (numbers: JP18K07545, JP19H04976, and JP21K07473) awarded to Y.O. and a JSPS KAKENHI grant (number: JP19K23377) awarded to Y.B.
Writer data
Affiliations
Contributions
YO conceived the study. YO and YB designed the report. YB, MN, HS, NN, CS, and YO conducted experiments, analyzed information, or contributed essential materials. YB and YO wrote the manuscript. All authors reviewed and revised the newspaper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open up Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, equally long as you give advisable credit to the original writer(due south) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other 3rd party material in this article are included in the commodity'south Artistic Commons license, unless indicated otherwise in a credit line to the textile. If material is not included in the article's Creative Eatables license and your intended use is not permitted past statutory regulation or exceeds the permitted employ, yous volition need to obtain permission directly from the copyright holder. To view a re-create of this license, visit http://creativecommons.org/licenses/past/four.0/.
Reprints and Permissions
About this article
Cite this commodity
Bouchekioua, Y., Nebuka, M., Sasamori, H. et al. Serotonin 5-HT2C receptor knockout in mice attenuates fear responses in contextual or cued but not compound context-cue fear workout. Transl Psychiatry 12, 58 (2022). https://doi.org/10.1038/s41398-022-01815-2
-
Received:
-
Revised:
-
Accustomed:
-
Published:
-
DOI : https://doi.org/10.1038/s41398-022-01815-2
Source: https://www.nature.com/articles/s41398-022-01815-2
0 Response to "Critique Review on Serotonin Engages an Anxiety and Fear-promoting Circuit in the Extended Amygdala"
Post a Comment